Investigator-Initiated Studies
At Madrigal, we support Investigator-Initiated Studies (IIS) that advance medical and scientific knowledge about therapies for MASH.
Please note that an IIS may also be known by other names that include and/or encompass the following:
Investigator-Initiated Trials (IIT), Investigator-Sponsored Trials (IST), Investigator Sponsored Research (ISR), Physician Initiated Trials (PIT) and External Research Programs (ERP).
We actively support external research initiatives through our IIS program, through which we provide financial support for clinical research projects designed, initiated and sponsored by independent researchers. These projects should focus on treatment, diagnosis and outcomes in alignment with Madrigal’s areas of interest, address evidence gaps and unmet needs and explore potential areas of innovation. Each proposal is carefully reviewed for its scientific merit to ensure it fits strategically with our areas of research interest. These reviews are conducted by global committees composed of Madrigal medical and scientific staff from relevant therapeutic areas.
At this time, Madrigal can only support investigator-initiated study requests from the United States, Germany and France.
IIS Areas of Interest
Natural History of MASH (Non-Interventional)
- With compensated cirrhosis
- Pediatric MASH
Treatment for MASH
- Native American populations
- Asian, African, Latin American or Hispanic populations
- Pre/post-bariatric surgery
- Ages 18-30 or over 60
Value and Impact of Treating MASH with Moderate to Advanced Fibrosis*
Emerging or Established Care Pathways*
General Out of Scope
- MASH patient registries or drug registries
- Other causes of liver steatosis and/or fibrosis
- Validation of machine-learning or AI tools
- Genome-Wide Association Study (GWAS)
*Proposals for these areas of interest will be considered for countries in EU where resmetirom is approved
Submit an IIS Concept Proposal
(U.S., Germany and France Only)
Create an account and begin the IIS concept application process
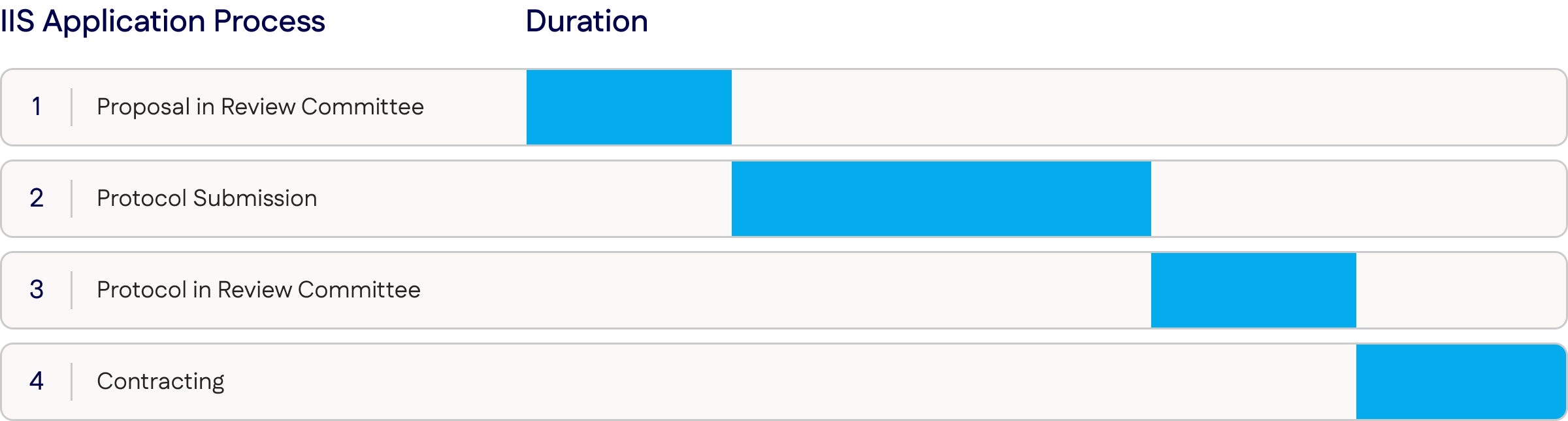
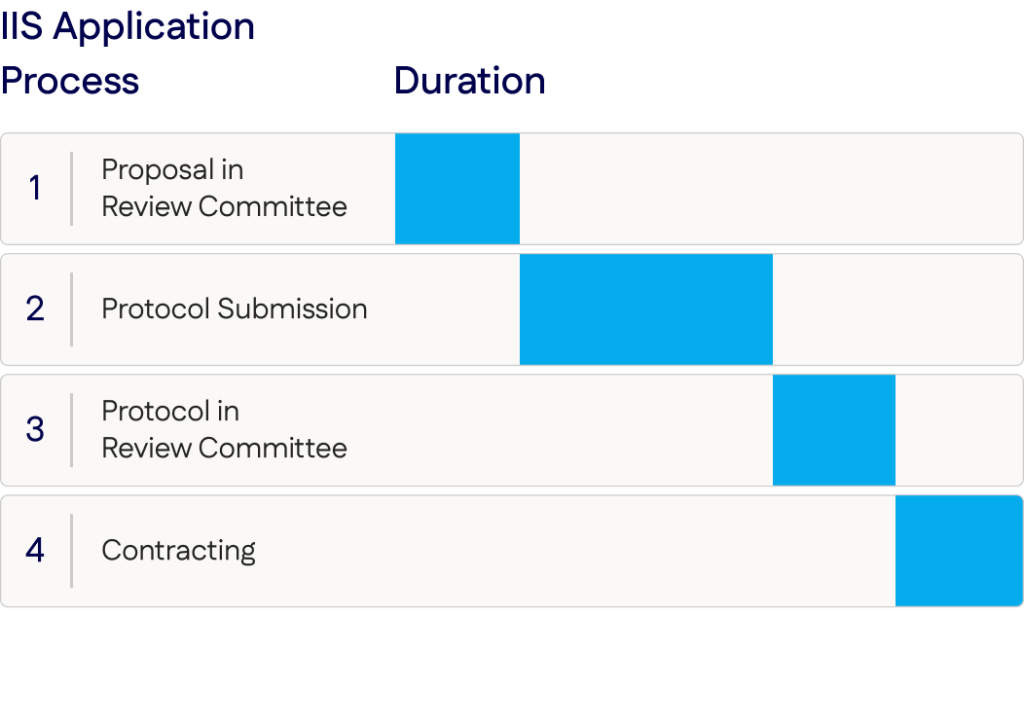
IIS Proposal Steps
your research proposal




Please note that an IIS application may be rejected at any stage within the review process
IIS Proposal Timing
The application portal will be open year-round for proposal submissions.
← Scroll horizontally to view more →
Frequently Asked Questions
What is an IIS?
- An IIS is a research project initiated, designed and sponsored by independent researchers or academic institutions
- Unlike studies led directly by pharmaceutical companies, IIS projects are conducted without the company’s direct involvement
- These studies focus on various aspects of treatment, diagnosis and outcomes related to the company’s products
- They are typically supported by the company through financial resources and aim to address evidence gaps and unmet needs and explore new areas of innovation
What process should I follow if I already have a fully developed study protocol?
- Initially, investigators must submit an IIS concept proposal and budget via the online application
- If Madrigal expresses interest in your proposal, you will be asked to submit a full study protocol
Can proposals that require drug supply be submitted to the portal?
- Madrigal can provide drug supply for certain approved IIS proposals. If your proposal includes a request for drug supply, please complete the appropriate section of the application